
Chemistry, 09.12.2021 19:40 2337911726
Consider the equations below.
4 equations. 1, Upper C Upper H Subscript 4 Baseline (g) right arrow Upper C (s) + 2 Upper H Subscript 2 Baseline (g) Delta H Subscript 1 Baseline = 74.6 kilojoules. 2, Upper C (s) + 2 Upper C l Subscript 2 Baseline (g) right arrow Upper C Upper Cl Subscript 4 Baseline (g) Delta H Subscript 2 Baseline = negative 95.7 kilojoules. 3, 2 Upper H Subscript 2 Baseline (g) + 2 Upper C l Subscript 2 Baseline (g) right arrow 4 Upper H Upper Cl (g) delta H Subscript 3 Baseline = negative 284.6 kilojoules. 4, Upper C Upper H Subscript 4 Baseline (g) + 4 Upper C l Subscript 2 Baselines (g) right arrow Upper C Upper C l Subscript 4 Baseline (g) + 4 Upper H Upper C L (g) Delta H 4 = negative 205.7 kilojoules.
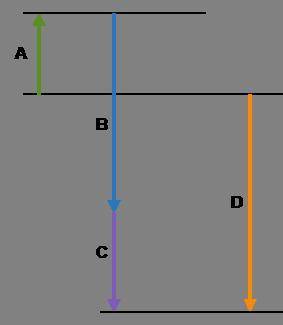
Complete the following based on the diagram.
Arrow A:
Arrow B:
Arrow C:
Arrow D:

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3


Chemistry, 22.06.2019 21:00
How many neutrons does an element have if its atomic number is 50 and its mass number is 166
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1
You know the right answer?
Consider the equations below.
4 equations. 1, Upper C Upper H Subscript 4 Baseline (g) right arrow...
Questions


Mathematics, 01.12.2019 23:31


Mathematics, 01.12.2019 23:31

Mathematics, 01.12.2019 23:31

Mathematics, 01.12.2019 23:31

Mathematics, 01.12.2019 23:31


Chemistry, 01.12.2019 23:31

Mathematics, 01.12.2019 23:31


English, 01.12.2019 23:31





Mathematics, 01.12.2019 23:31

English, 01.12.2019 23:31


Mathematics, 01.12.2019 23:31



