A + B
AB
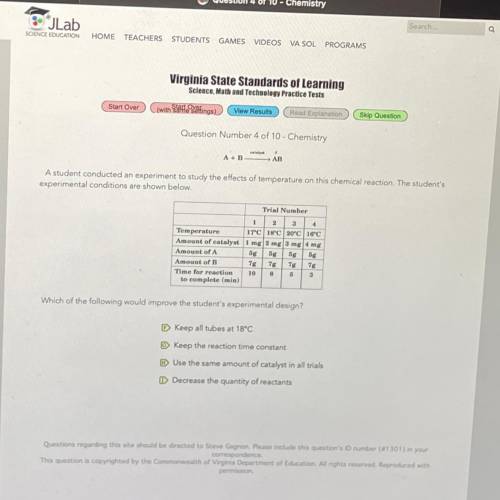
A student conducted an experiment to study the effects of temperature on this chemi...

Chemistry, 09.12.2021 01:00 oscarsanchez1530
A + B
AB
A student conducted an experiment to study the effects of temperature on this chemical reaction. The student's
experimental conditions are shown below.
Trial Number
2 3 4
Temperature 17 18°C 20°C 16°C
Amount of catalyst 1 mg 2 mg 3 mg 4 mg
Amount of A
5g 5g 5g bg
Amount of B
7g 7g 7g 7g
Time for reaction 10 8 5 3
to complete (min)
Which of the following would improve the student's experimental design?
Keep all tubes at 18°C
Keep the reaction time constant
Use the same amount of catalyst in all trials
Decrease the quantity of reactants

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 18:00
Which three statements represent the benefits of performing experiments using computer simulations?
Answers: 2

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3

Chemistry, 22.06.2019 19:30
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2
You know the right answer?
Questions



Social Studies, 24.03.2020 23:58

Social Studies, 24.03.2020 23:58






Mathematics, 24.03.2020 23:58

Mathematics, 24.03.2020 23:58




Mathematics, 24.03.2020 23:58







