Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:20
Part b: study of equilibrium on solubility: mg(oh)2(s) ⇌ mg2+(aq) + 2 oh–(aq) cloudy clear (pink) 7. a. b. 8. a. b. 9. 10. 11. 12. when adding concentrated hydrochloric acid, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 7a. you should indicate which ion was added to or removed from the equilibrium mixture. when adding edta, how did the appearance of the equilibrium mixture change? the change in appearance indicated a shift in the point of equilibrium. in which direction did the equilibrium shift? (l) left (r) right explain your answer to question 8a. you should indicate which ion was added to or removed from the equilibrium mixture. upon heating in which direction is the equilibrium shifting? upon cooling in which direction is the equilibrium shifting? is the forward reaction a. endothermic explain your answers to questions 9, 10, and 11. (l) left (r) right (l) left (r) right b. exothermic
Answers: 1

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3

Chemistry, 22.06.2019 14:30
What type(s) of intermolecular forces are expected between ch3ch2cooh molecules? dipole forces, induced dipole forces, hydrogen bonding
Answers: 1

You know the right answer?
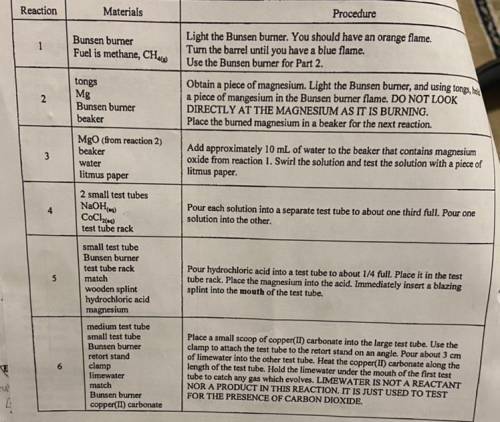
Can someone please help me write the 6 balanced chemical equation? I really need help I need to have...
Questions

Mathematics, 05.01.2021 03:30

Mathematics, 05.01.2021 03:30

Mathematics, 05.01.2021 03:30


History, 05.01.2021 03:30




Mathematics, 05.01.2021 03:30

Mathematics, 05.01.2021 03:30

Mathematics, 05.01.2021 03:30


Mathematics, 05.01.2021 03:30


History, 05.01.2021 03:30

Mathematics, 05.01.2021 03:30

Mathematics, 05.01.2021 03:30

English, 05.01.2021 03:30

English, 05.01.2021 03:30

Chemistry, 05.01.2021 03:30