
Chemistry, 01.12.2021 08:30 vickydaiberg
Calculate the reaction rate if [Cl2] = 0.012M, [O2] = 0.250M and the rate constant is 2.54x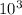
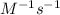 at 25C.
at 25C.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Calculate the change in entropy if br2(l) is converted into gaseous br atoms. s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 2

Chemistry, 21.06.2019 20:10
Why is the vapor pressure of a warm lake higher than the vapor pressure of a cold lake? o a. warm water has a greater heat of vaporization. ob. warm water evaporates more quickly. cool water evaporates more quickly. od. cool water has a greater heat of vaporization.
Answers: 1

Chemistry, 23.06.2019 00:20
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 15:40
Which functions of water in living systems would still be possible if water was not polar and did not form hydrogen bonds? check all that apply. climate regulation dissolving ionic compounds for biological reactions providing body support by exerting pressure on cell walls providing body support through buoyancy transport of nutrients within organisms temperature regulation in many organisms
Answers: 3
You know the right answer?
Calculate the reaction rate if [Cl2] = 0.012M, [O2] = 0.250M and the rate constant is 2.54x at 25C....
Questions


Mathematics, 22.01.2021 17:50



Social Studies, 22.01.2021 17:50

Social Studies, 22.01.2021 17:50

Computers and Technology, 22.01.2021 17:50

Mathematics, 22.01.2021 17:50

Arts, 22.01.2021 17:50



Mathematics, 22.01.2021 17:50










