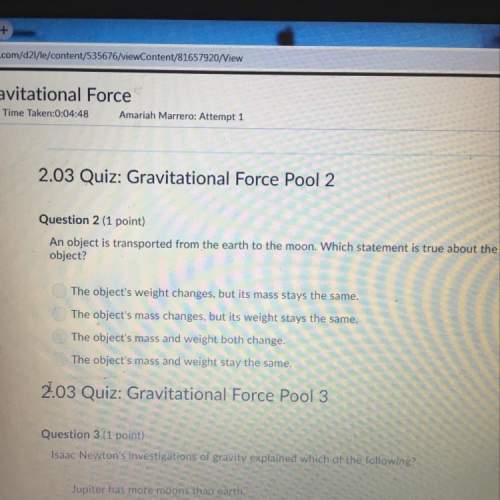
Ssignment (1) Nitrobenzene has a dipole moment of 4.2 x 10-9 esu cm while phenol has a value of only 1.7 x 10-8 esu cm. However, nitrobenzene is soluble only to the extent of 0.0155mole/kg in water while phenol is soluble to the extent of 0.95mole/kgat 20C. explain this phenomenon.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:50
Which best describes why nh4+ can form an ionic bond with cl-?
Answers: 3


Chemistry, 22.06.2019 06:00
If you burn 10 kilograms of wood in a fire (combustion) what is the weight of the products after the fire has finished burning the wood?
Answers: 3

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3
You know the right answer?
Ssignment (1) Nitrobenzene has a dipole moment of 4.2 x 10-9 esu cm while phenol has a value of only...
Questions


Mathematics, 08.12.2020 03:10


Mathematics, 08.12.2020 03:10




Mathematics, 08.12.2020 03:10

Mathematics, 08.12.2020 03:10

Mathematics, 08.12.2020 03:10

Health, 08.12.2020 03:10


History, 08.12.2020 03:10

Biology, 08.12.2020 03:10

Mathematics, 08.12.2020 03:10

Mathematics, 08.12.2020 03:10




History, 08.12.2020 03:10