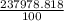Chemistry, 04.11.2021 09:30 arushiverma555
Uranium has three common isotopes: U-238 (99.2745%), U-235 (0.7200%), U-234 (0.0055%). Calculate the average atomic mass of uranium. (Round to two decimal places)

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 16:30
An atom with 7 protons, 6 neutrons, and 7 electrons has an atomic mass of amu. (enter a whole number.) numerical answers expected! answer for blank 1:
Answers: 3

Chemistry, 22.06.2019 18:30
How many moles of lead are in 1.50 x 10^12 atoms of lead? could you explain the answer as well and not just give it to me i am refreshing for finals and i need to know how to do it
Answers: 3
You know the right answer?
Uranium has three common isotopes: U-238 (99.2745%), U-235 (0.7200%), U-234 (0.0055%). Calculate the...
Questions

English, 07.01.2021 06:30

Mathematics, 07.01.2021 06:30

Chemistry, 07.01.2021 06:30

Mathematics, 07.01.2021 06:30


Mathematics, 07.01.2021 06:30


Social Studies, 07.01.2021 06:30

History, 07.01.2021 06:30


Mathematics, 07.01.2021 06:30

Mathematics, 07.01.2021 06:30

Mathematics, 07.01.2021 06:30


Mathematics, 07.01.2021 06:30

History, 07.01.2021 06:30

Mathematics, 07.01.2021 06:30

Advanced Placement (AP), 07.01.2021 06:30

English, 07.01.2021 06:30