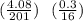Chemistry, 10.10.2021 21:40 skyemichellec
A compound of mercury and oxygen is heated in order to decompose the compound. A 4.08 grams sample of mercury oxide upon heating gives off oxygen gas. After heating the mass of mercury metal left behind was 3.78 grams. Calculate the empirical formula of the compound
My and: 1 O2 to 2 Hg ratio but I don't know if it's correct and if so if I should write it as
2HgO > 2Hg + O2 or just 2Hg + O2
Please someone who understands help.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:30
Infants born with severe respiratory problems are sometimes given liquid ventilation: they breathe a liquid that can dissolve more oxygen than air can hold. one of these liquids is a fluorinated compound, cf3(cf2)7br. the solubility of oxygen in this liquid is 66 mlo2 per 100 ml liquid. in contrast, air is 21 % oxygen by volume. calculate the moles of o2 present in an infant's lungs (volume: 12 ml ) if the infant takes a full breath of air. assume a pressure of 1 atm in the lungs.
Answers: 1

Chemistry, 22.06.2019 12:00
From the options provided for each element below, choose the properties that it may have based on its location in the periodic table fluorine (f): highly reactive nonmetal shiny a conductor
Answers: 1

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 23.06.2019 00:00
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3
You know the right answer?
A compound of mercury and oxygen is heated in order to decompose the compound. A 4.08 grams sample o...
Questions














Mathematics, 25.03.2020 06:27


Biology, 25.03.2020 06:27

English, 25.03.2020 06:27

History, 25.03.2020 06:27