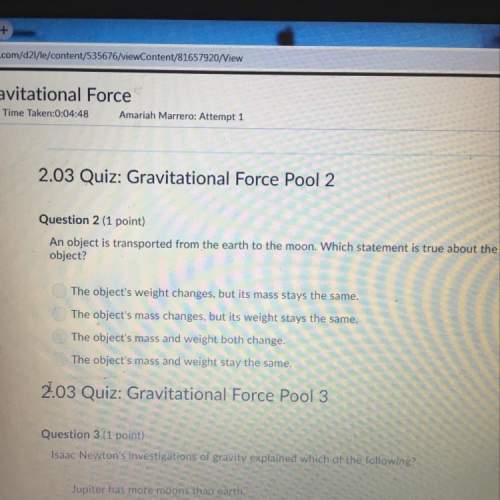
Chemistry, 05.10.2021 14:00 keniaguevara32
The empirical formula of an insecticide is found to be CHCl, and its molar mass is 290.8 g/mol. How many atoms of carbon are found in a molecule of the insecticide?

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1


Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
The empirical formula of an insecticide is found to be CHCl, and its molar mass is 290.8 g/mol. How...
Questions


History, 09.12.2020 02:00


Mathematics, 09.12.2020 02:00

Mathematics, 09.12.2020 02:00

Mathematics, 09.12.2020 02:00

Chemistry, 09.12.2020 02:10

Mathematics, 09.12.2020 02:10

Mathematics, 09.12.2020 02:10


History, 09.12.2020 02:10

History, 09.12.2020 02:10



English, 09.12.2020 02:10


Social Studies, 09.12.2020 02:10

Mathematics, 09.12.2020 02:10


Mathematics, 09.12.2020 02:10