
Chemistry, 04.10.2021 20:30 Victoriag2626
50 Points Help
1) A block of aluminum occupies a volume of 15.0 mL and mass 40.5 g. What is its density?
2) Mercury metal is poured into a graduated cylinder that holds exactly 22.5 mL. The mercury used to fill the cylinder mass 306.0 g. From this information, calculate the density of mercury.
3) What is the mass of the ethyl alcohol that exactly fills a 200.0 mL container? The density of ethyl alcohol is 0.789 g/mL.
4) A rectangular block of copper metal weighs 1896 g. The dimensions of the block are 8.4 cm by 5.5 cm by 4.6 cm. From this data, what is the density of copper? ( hint find the volume first: volume of rectangle = Hight x width x length)
5) Calculate the density of sulfuric acid if 35.4 mL of the acid mass 65.14 g.

Answers: 1


Another question on Chemistry

Chemistry, 23.06.2019 00:00
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3

Chemistry, 23.06.2019 10:30
4al + 3o2 → 2al2o3 what does the "3" in front of o2 stand for? a) it indicates that there are 5 oxygen atoms after you add the coefficient and the subscript. b) it indicates that that there are are total of 6 oxygen atoms all bonded together as a single molecule. c) it indicates that there are 3 oxygen molecules chemically bonded to each other in the reaction. d) it indicates that there are 3 separate oxygen molecules in the reaction.
Answers: 2

Chemistry, 23.06.2019 11:50
How many moles of an ideal gas would occupy a 25.0 liter container when the temperature is 295 k and the pressure is 0.850 atm?
Answers: 2

Chemistry, 23.06.2019 16:30
Which temperature scale measures water boiling at 100 degrees?
Answers: 2
You know the right answer?
50 Points Help
1) A block of aluminum occupies a volume of 15.0 mL and mass 40.5 g. What is its de...
Questions

English, 01.03.2020 22:17

Mathematics, 01.03.2020 22:18


History, 01.03.2020 22:18


Mathematics, 01.03.2020 22:19


Computers and Technology, 01.03.2020 22:23

Mathematics, 01.03.2020 22:24

Biology, 01.03.2020 22:25

Mathematics, 01.03.2020 22:25


Mathematics, 01.03.2020 22:25

Mathematics, 01.03.2020 22:25

Mathematics, 01.03.2020 22:25

Chemistry, 01.03.2020 22:25


Mathematics, 01.03.2020 22:26

English, 01.03.2020 22:26

Physics, 01.03.2020 22:26

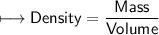







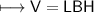
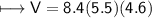
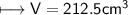

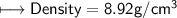

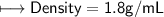

 when dealing with a solid.
when dealing with a solid. or
or 



