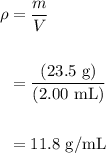Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 16:20
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.
Answers: 2


Chemistry, 21.06.2019 22:30
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 09:10
Select the correct answer from each drop-down menu.describe what happens to a carbon-11 atom when it undergoes positron emission.the decay of a carbon-11 atom _1_, and this causes it to emit _2_.options for 1: > changes a neutron into a proton> changes a proton into a neutron> is hit with a neutron> reconfigures its protons and neutronsoptions for 2: > a negatively charged electron-sized particle> a positively charged election-sized particle> two atoms and several neutrons> two neutrons and two protons
Answers: 3
You know the right answer?
A student massed an unknown sample and found it to be 23.5g. The volume of the sample is 2.00mL. Cal...
Questions

Mathematics, 04.07.2019 21:30


Biology, 04.07.2019 21:30

Mathematics, 04.07.2019 21:30

Mathematics, 04.07.2019 21:30


History, 04.07.2019 21:30



Mathematics, 04.07.2019 21:30

Advanced Placement (AP), 04.07.2019 21:30

Social Studies, 04.07.2019 21:30

Mathematics, 04.07.2019 21:30

Mathematics, 04.07.2019 21:30

History, 04.07.2019 21:30


Biology, 04.07.2019 21:30

Mathematics, 04.07.2019 21:30


History, 04.07.2019 21:30