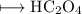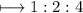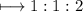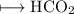Chemistry, 07.09.2021 07:50 leannaadrian
The empirical formula for the compound having the formula H C204

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 22.06.2019 09:00
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 11:30
If we compare and contrast electromagnetic waves with sound waves, all but one statement is true. that is a) sound waves require a medium to travel while electromagnetic waves do not. b) electromagnetic waves can travel through the vacuum of space while sound waves cannot. c) electromagnetic waves must have a medium in which to travel, but sound waves can travel anywhere. eliminate d) sound waves must bounce off of matter in order to travel while electromagnetic waves do not require matter to be present.
Answers: 3

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1
You know the right answer?
The empirical formula for the compound having the formula H C204...
Questions

German, 22.01.2021 18:20

English, 22.01.2021 18:20

Mathematics, 22.01.2021 18:20

Chemistry, 22.01.2021 18:20




Mathematics, 22.01.2021 18:20

Chemistry, 22.01.2021 18:20




Mathematics, 22.01.2021 18:20

Mathematics, 22.01.2021 18:20


Mathematics, 22.01.2021 18:20


Mathematics, 22.01.2021 18:20


Chemistry, 22.01.2021 18:20