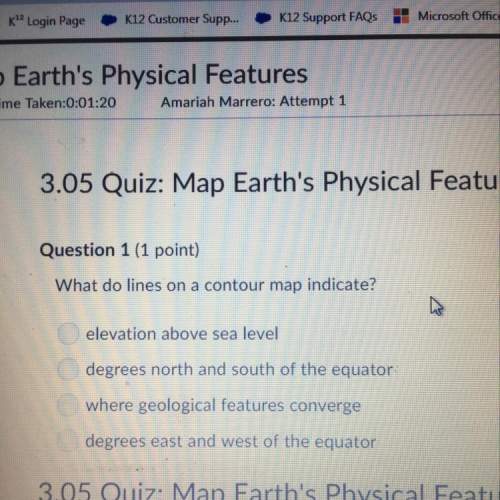
Chemistry, 07.09.2021 01:20 savannahsharp4463
160g of aluminum is burned in excess air to produce aluminum oxide, Al2O3(s). When
the reaction is complete, 260g of solid aluminum oxide has been produced.
Write a balanced equation for the reaction. (1 mark)
Calculate the theoretical yield. (2 marks)
.
Calculate the percentage yield. (2 marks)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
The drawing represents the movement of particles in a substance. what changes of state can this substance undergo
Answers: 1

Chemistry, 22.06.2019 04:30
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1

Chemistry, 22.06.2019 06:00
One of the few xenon compounds that form is cesium xenon heptafluoride (csxef7). how many moles of csxef7 can be produced from the reaction of 13.0 mol cesium fluoride with 12.5 mol xenon hexafluoride? csf(s) + xef6(s) csxef7(s)
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3
You know the right answer?
160g of aluminum is burned in excess air to produce aluminum oxide, Al2O3(s). When
the reaction is...
Questions


Biology, 20.09.2020 05:01


Mathematics, 20.09.2020 05:01






Computers and Technology, 20.09.2020 05:01


Mathematics, 20.09.2020 05:01




Computers and Technology, 20.09.2020 05:01


Mathematics, 20.09.2020 05:01