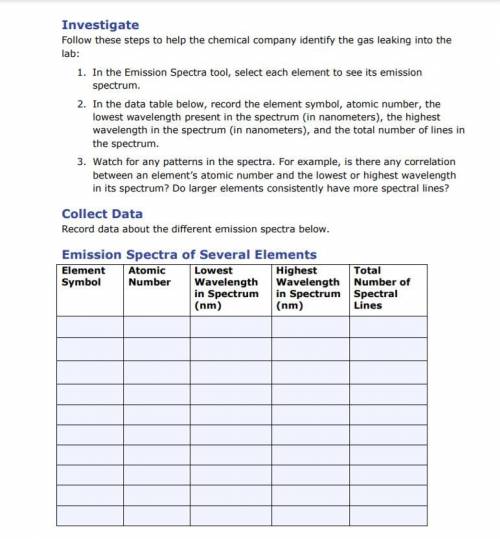
1. In the Emission Spectra tool, select each element to see its emission
spectrum.
2. In th...

1. In the Emission Spectra tool, select each element to see its emission
spectrum.
2. In the data table below, record the element symbol, atomic number, the
lowest wavelength present in the spectrum (in nanometers), the highest
wavelength in the spectrum (in nanometers), and the total number of lines in
the spectrum.
3. Watch for any patterns in the spectra. For example, is there any correlation
between an element’s atomic number and the lowest or highest wavelength
in its spectrum? Do larger elements consistently have more spectral lines?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:00
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 22:30
Gusing the milligrams of ascorbic acid you entered above, the ratio of total sample volume to aliquot volume, and the total milligrams of the vitamin c tablet that you dissolved, calculate the mass of ascorbic acid in the vitamin c tablet for each trial. do this by scaling up to find the amount (mg) of ascorbic acid in your 250 ml flask. enter your calculated mass of ascorbic acid in the vitamin c tablet, for each trial. be sure to enter your calculated mass in the corresponding order that you entered your milligrams of ascorbic acid. the milligrams of ascorbic acid you entered for entry #1 previously should correspond to the mass of ascorbic acid that you enter for entry #1 here.
Answers: 1

You know the right answer?
Questions


Chemistry, 18.05.2021 19:20

Mathematics, 18.05.2021 19:20

Mathematics, 18.05.2021 19:20

History, 18.05.2021 19:20



History, 18.05.2021 19:20

Mathematics, 18.05.2021 19:20







Mathematics, 18.05.2021 19:20

History, 18.05.2021 19:20

History, 18.05.2021 19:20




