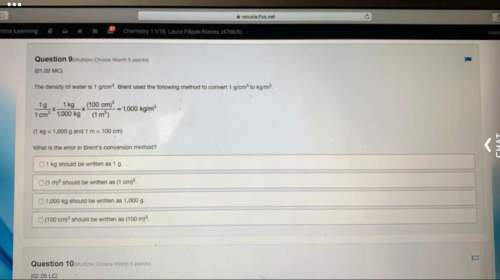
Chemistry, 25.08.2021 16:20 chasewilkinson123
Why the volume of a gas is not measured immediately after it is collected?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Hot air balloons float in the air because of the difference in density between cold and hot air. in this problem, you will estimate the minimum temperature the gas inside the balloon needs to be, for it to take off. to do this, use the following variables and make these assumptions: the combined weight of the pilot basket together with that of the balloon fabric and other equipment is w. the volume of the hot air inside the balloon when it is inflated is v. the absolute temperature of the hot air at the bottom of the balloon is th (where th> tc). the absolute temperature of the cold air outside the balloon is tc and its density is ďc. the balloon is open at the bottom, so that the pressure inside and outside the balloon is the same. as always, treat air as an ideal gas. use g for the magnitude of the acceleration due to gravity.
Answers: 1

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 18:30
Which of the following words describe the reality that the universe looks the same from various perspective
Answers: 3

Chemistry, 23.06.2019 03:00
Abaker touches a pie right after taking it out of the oven. which statement best explains why the pie feels hot?
Answers: 2
You know the right answer?
Why the volume of a gas is not measured immediately after it is collected?...
Questions


Spanish, 28.01.2020 16:56

Computers and Technology, 28.01.2020 16:56

Health, 28.01.2020 16:56

English, 28.01.2020 16:56




English, 28.01.2020 16:57

History, 28.01.2020 16:57


Business, 28.01.2020 16:57

English, 28.01.2020 16:57


History, 28.01.2020 16:57




Physics, 28.01.2020 16:57