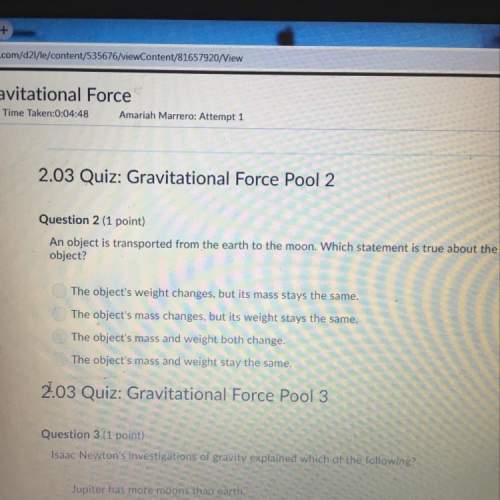
Chemistry, 16.08.2021 22:00 niceguy1997
the energy required for the complete removal of 1 mol of electrons from 1 mole of gaseous atoms or ions is called energy. For a neutral element this energy is a measure of how easily the element forms an ion with a charge.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2

Chemistry, 22.06.2019 10:50
An atom of lithium-7 has an equal number of(1) electrons and neutrons(2) electrons and protons(3) positrons and neutrons(4) positrons and protons
Answers: 2

Chemistry, 22.06.2019 14:50
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of tthe table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 9 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3
You know the right answer?
the energy required for the complete removal of 1 mol of electrons from 1 mole of gaseous atoms or i...
Questions

Physics, 18.09.2019 05:20


History, 18.09.2019 05:20


Mathematics, 18.09.2019 05:20



History, 18.09.2019 05:20


Biology, 18.09.2019 05:20

Mathematics, 18.09.2019 05:20


Mathematics, 18.09.2019 05:20


History, 18.09.2019 05:20

Mathematics, 18.09.2019 05:20

Biology, 18.09.2019 05:30

English, 18.09.2019 05:30

Social Studies, 18.09.2019 05:30

Social Studies, 18.09.2019 05:30