1. Based on the following balanced equation:
2 H2 + O2 + 2H2O
a. How many moles of O2 are req...

Chemistry, 15.08.2021 01:00 dirtridersteve65
1. Based on the following balanced equation:
2 H2 + O2 + 2H2O
a. How many moles of O2 are required to react completely with 2.00 moles of H2?
1.00 1.50 2.00 2.50 3.00 3.50 4.00 4.50 5.00
b. How many moles of H2O are produced after complete reaction of 2.00 moles of H2?
1.00 1.50
2.00 2.50 3.00 3.50 4.00 4.50 5.00

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 10:40
Asolid that forms and separates from a liquid mixture is called
Answers: 2

Chemistry, 23.06.2019 03:00
Select the correct answer. wax is a nonpolar substance. in which type of substance is it the most soluble?
Answers: 1


Chemistry, 23.06.2019 09:00
Which factor is likely to impact the possible number of compounds? presence of unlimited number of elements in the periodic table the inability of atoms to align perfectly with other atoms the ability of all elements to react with every other element all elements being equally reactive
Answers: 2
You know the right answer?
Questions

Social Studies, 10.01.2020 20:31





History, 10.01.2020 20:31



Mathematics, 10.01.2020 20:31

Mathematics, 10.01.2020 20:31


History, 10.01.2020 20:31


Law, 10.01.2020 20:31


Mathematics, 10.01.2020 20:31

Mathematics, 10.01.2020 20:31

Mathematics, 10.01.2020 20:31


Biology, 10.01.2020 20:31

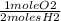 = 1 mole O2
= 1 mole O2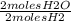 = 2 moles H20
= 2 moles H20

