Chemistry, 08.08.2021 06:00 destinywashere101
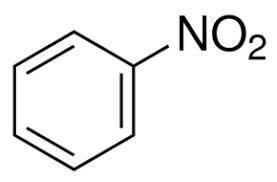
Suppose 'A' is a liquid aromatic compound with molecular weight 78 and burns with sooty flame. a. Give the name of the compound 'A' b. write the molecular structure of 'A' C. What is the product when 'A' is treated with ? i. conc. HNO3 with conc. H2SO4 as catalyst ii. Halogen (cl2)in presence of sunlight and mention the use of the product obtained

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 12:30
Which statement is true about this reaction? 14n+1h 15o it is a practical source of energy on earth. it occurs only outside the solar system. its product is heavier than each of its reactants. it shows the critical mass of an element.
Answers: 2

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2

Chemistry, 22.06.2019 16:50
Answer asap need it by wednesday morning calculate the ph of 0.02m hcl best answer will be brainliest
Answers: 1
You know the right answer?
Suppose 'A' is a liquid aromatic compound with molecular weight 78 and burns with sooty flame. a. Gi...
Questions




Social Studies, 20.07.2019 20:00





Physics, 20.07.2019 20:00

Physics, 20.07.2019 20:00





Mathematics, 20.07.2019 20:00





Mathematics, 20.07.2019 20:00