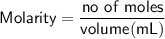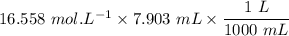Sally has constructed a concentration cell to measure Ksp for MCln. She constructs the cell by adding 2 mL of 0.05 M M(NO3)n to one compartment of the microwell plate. She then makes a solution of MCln by adding KCl to M(NO3)n. She adds 7.903 mL of the resulting mixture to a second compartment of the microwell plate. Sally knows n = +2. She has already calculated [Mn+] in the prepared MCln solution using the Nernst equation. [Mn+] = 8.279 M
Required:
How many moles of [Cl-] must be dissolved in that compartment?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1

Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2

Chemistry, 22.06.2019 19:30
Astudent conducts an experiment to determine how the amount of water given to a plant affects its growth. what is the independent variable for this experiment?
Answers: 1
You know the right answer?
Sally has constructed a concentration cell to measure Ksp for MCln. She constructs the cell by addin...
Questions



Social Studies, 16.02.2022 14:00

Physics, 16.02.2022 14:00



Mathematics, 16.02.2022 14:00



History, 16.02.2022 14:00


SAT, 16.02.2022 14:00

Chemistry, 16.02.2022 14:00




English, 16.02.2022 14:00


English, 16.02.2022 14:00

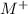 reacted with Cl⁻ to form
reacted with Cl⁻ to form 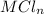 i.e. the compound formed is
i.e. the compound formed is 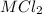 .
.![[M^+]](/tpl/images/1402/0086/bbccd.png) = 8.279 M
= 8.279 M![[Cl^-]](/tpl/images/1402/0086/0726e.png) = 2 × 8.279 M
= 2 × 8.279 M