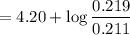Chemistry, 28.07.2021 03:00 nauticatyson9
An analytical chemist is titrating of a solution of benzoic acid with a solution of . The of benzoic acid is . Calculate the pH of the acid solution after the chemist has added of the solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of solution added.

Answers: 2


Another question on Chemistry



Chemistry, 22.06.2019 10:30
Balance and in which category does it fit in? single or double displacement or synthesis or decomposition? (a) k2 o → k + o2 (b) na + i2 → nai (c) cu(no3 )2 + naoh → cu(oh)2 + nano3 (d) kclo3 → kcl + o2 (e) ca(no3 )2 + hbr → cabr2 + hno3 (f) sn(oh)2 → sno + h2 o (g) p4 + n2 o → p4 o6 + n2 (h) fe + al2 (so4 )3 → feso4 + al (i) alcl3 + na2 co3 → al2 (co3 )3 + nacl (j) c3 h6 + o2 → co2 + h2 o
Answers: 1

Chemistry, 22.06.2019 11:00
Which element would mostly likely have an electron affinity measuring closest to zero
Answers: 3
You know the right answer?
An analytical chemist is titrating of a solution of benzoic acid with a solution of . The of benzoic...
Questions


Social Studies, 17.07.2021 14:50

Mathematics, 17.07.2021 14:50


English, 17.07.2021 14:50


Computers and Technology, 17.07.2021 14:50


Chemistry, 17.07.2021 14:50


Mathematics, 17.07.2021 15:00

Mathematics, 17.07.2021 15:00

Mathematics, 17.07.2021 15:00

English, 17.07.2021 15:00

Geography, 17.07.2021 15:00

Social Studies, 17.07.2021 15:00


Mathematics, 17.07.2021 15:00

Arts, 17.07.2021 15:00

Arts, 17.07.2021 15:00

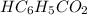 with a 0.3600 M solution of KOH. The
with a 0.3600 M solution of KOH. The 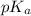 of benzoic acid is 4.20. Calculate the pH of the acid solution after the chemist has added 232.0 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added.
of benzoic acid is 4.20. Calculate the pH of the acid solution after the chemist has added 232.0 mL of the KOH solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of KOH solution added.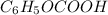
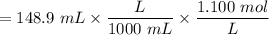
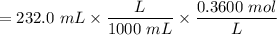
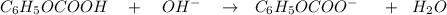
![$C_6H_5OCOOH, [C_6H_5OCOOH]$](/tpl/images/1400/7778/75042.png)
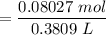
![$C_6H_5OCOO^- , [C_6H_5OCOO^-] =\frac{0.08352 \ mol}{0.3809 \ L}](/tpl/images/1400/7778/c3e04.png)
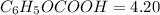
![$pH = pK_a + \log \frac{[C_6H_5OCOO^-]}{[C_6H_5OCOOH]}](/tpl/images/1400/7778/ac810.png)