
Chemistry, 27.07.2021 06:30 augestfaith
How many milliliters of 0.204 Mol KMnO4 are needed to react with 3.24 g of iron(II) sulfate, FeSO4? The reation is as folows. 10FeSO4(aq) + 2 KMnO4(aq) = 5Fe2(SO4)3(aq) + 2MnSO4(aq) + K2SO4(aq) + 8H2O(l)
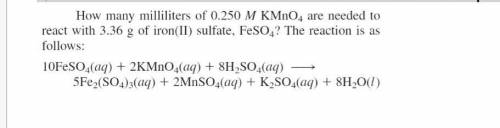

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:00
This flow chart shows the amount of energy that is emitted by each type of light. ultraviolet > blue light > yellow light > red light (maximum energy) (minimum energy) in an experiment, shining which type of light on a strip of metal would be least likely to produce the photoelectric effect? ultraviolet light dim blue light bright red light bright yellow light
Answers: 2

Chemistry, 22.06.2019 08:30
Which common material is an example of a polymer? (25 pts) a. steel b. plastic c. petroleum d. rubbing alcohol
Answers: 2

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 3

Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1
You know the right answer?
How many milliliters of 0.204 Mol KMnO4 are needed to react with 3.24 g of iron(II) sulfate, FeSO4?...
Questions

History, 18.08.2019 09:30


Mathematics, 18.08.2019 09:30

English, 18.08.2019 09:30


Health, 18.08.2019 09:30

Mathematics, 18.08.2019 09:30

English, 18.08.2019 09:30

Mathematics, 18.08.2019 09:30



Health, 18.08.2019 09:30


Social Studies, 18.08.2019 09:30




History, 18.08.2019 09:30




