
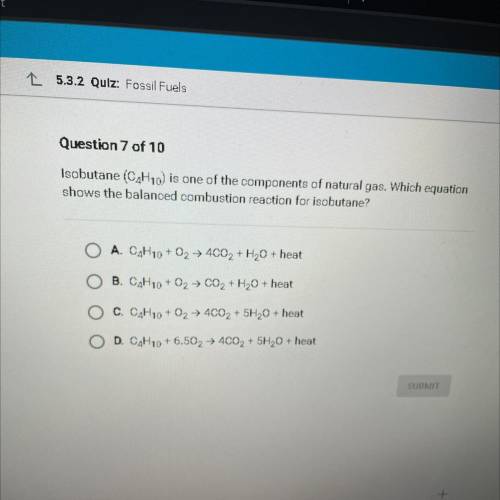
Isobutane (C4H10) is one of the components of natural gas. Which equation
shows the balanced combustion reaction for isobutane?
A. C4H10 + O2 + 4CO2 + H2O + heat
B. C4H10 + O2 + CO2 + H2O + heat
C. C4H10 + O2 + 4C02 + 5H20 + heat
D. C4H10 +6.502 → 4002 + 5H20 + heat
SUBMIT

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:20
Which statement accurately describes the relationship between air pressure, air density, or altitude? as altitude increases, pressure increases.as altitude increases, air density increases.air pressure and density are lowest at sea level.denser air exerts more pressure than less dense air.
Answers: 2


Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 08:30
If i initially have a gas at a pressure of 12 atm, a volume of 23 liters, and a temperature of 200 k, and then i raise the pressure to 14 atm and increase the temperature to 300 k, what is the new volume of the gas?
Answers: 2
You know the right answer?
Isobutane (C4H10) is one of the components of natural gas. Which equation
shows the balanced combus...
Questions

Business, 25.01.2020 03:31





Social Studies, 25.01.2020 03:31




Computers and Technology, 25.01.2020 03:31

Mathematics, 25.01.2020 03:31

Computers and Technology, 25.01.2020 03:31






Mathematics, 25.01.2020 03:31




