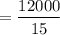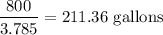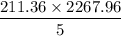A clean-burning automobile engine emits about 5 lb of C atoms in the form of CO2 molecules for every gallon of gasoline it consumes. The average American car is driven about 12,000 miles per year. Using this information, check the statement that the average American car releases its own weight in carbon into the atmosphere each year. List the assumptions you make to solve this problem.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Chemistry, 22.06.2019 23:00
What prefix multiplier is appropriate for reporting a measurement of 5.57 ×10−5 m?
Answers: 1
You know the right answer?
A clean-burning automobile engine emits about 5 lb of C atoms in the form of CO2 molecules for every...
Questions

English, 24.09.2019 05:30

Mathematics, 24.09.2019 05:30



Business, 24.09.2019 05:30


English, 24.09.2019 05:30



Biology, 24.09.2019 05:30

Mathematics, 24.09.2019 05:30

Social Studies, 24.09.2019 05:30



Mathematics, 24.09.2019 05:30


Social Studies, 24.09.2019 05:30

History, 24.09.2019 05:30

Mathematics, 24.09.2019 05:30

History, 24.09.2019 05:30