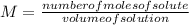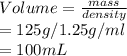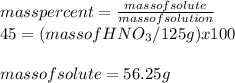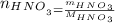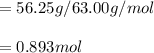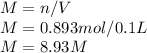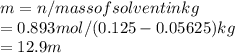Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:00
The study of witch tree monkeys feed in is part of the science life
Answers: 1

Chemistry, 22.06.2019 00:30
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2

Chemistry, 22.06.2019 02:30
You have a sample of a gas that occupies a volume of 17ml at -111 degrees celsius. what volume does the sample occupy at 88 degrees celsius? show all work asap
Answers: 3

Chemistry, 22.06.2019 02:50
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2
You know the right answer?
Calcular la m y M de una concentración Porcentual 45% m/m, la cual tiene una D solución 1.25 g/ml y...
Questions




Chemistry, 13.08.2019 02:10

Geography, 13.08.2019 02:10



Mathematics, 13.08.2019 02:10




Physics, 13.08.2019 02:10

Mathematics, 13.08.2019 02:10



Geography, 13.08.2019 02:10