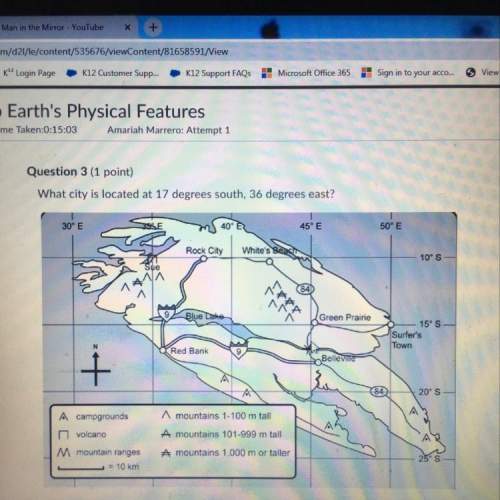
Chemistry, 20.07.2021 03:10 DWilson1234
Given the following reaction:
CO (g) + 2 H2(g) <==> CH3OH (g)
In an experiment, 0.42 mol of CO and 0.42 mol of H2 were placed in a 1.00-L reaction vessel. At equilibrium, there were 0.29 mol of CO remaining. Keq at the temperature of the experiment is .
A) 2.80
B) 0.357
C) 14.5
D) 17.5
E) none of the above

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:30
If 200.0g of copper(ll) sulfate react with an excess of zinc metal, what is the theoretical yield of copper
Answers: 1

Chemistry, 22.06.2019 00:00
1) these are barrel shaped microtubules in most animal cells, that organize the spindles during cell division
Answers: 1

Chemistry, 22.06.2019 08:30
Which metal exist in liquid state and can be cut with knife ?
Answers: 2

Chemistry, 22.06.2019 21:50
What is a main difference between a mixture and a pure substance? a mixture is only a liquid, but a pure substance can be in any state.a mixture looks the same throughout, but a pure substance does not.1 a mixture can vary in composition, but a pure substance has a set composlo a mixture can be made up of a single compound, but a pure substance car
Answers: 2
You know the right answer?
Given the following reaction:
CO (g) + 2 H2(g) <==> CH3OH (g)
In an experiment, 0.42 mo...
In an experiment, 0.42 mo...
Questions


History, 19.02.2020 05:23




English, 19.02.2020 05:24





Computers and Technology, 19.02.2020 05:24

History, 19.02.2020 05:24




Mathematics, 19.02.2020 05:25

Mathematics, 19.02.2020 05:25

Social Studies, 19.02.2020 05:25