Chemistry, 19.07.2021 17:40 gatorlove00
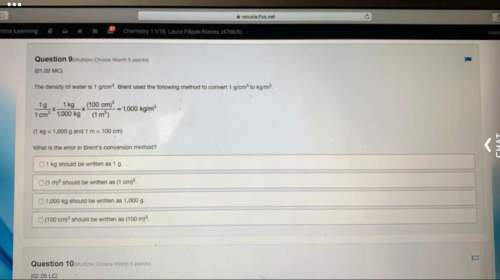
(1) A certain quantity of sodium tetraborate was weighed accurately and placed in a 250 ml volumetric flask. (ii) 25 mL of this stock solution was then titrated with the hydrochloric acid using bromocresol green as an indicator. If the average titre value obtained for this titration was 10.5 mL and the concentration of hydrochloric acid was determined to be 0.4 M, calculate the initial mass of sodium tetraborate that was weighed out.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which part of a feedback mechanism is able to monitor the conditions outside of cells and usually uses nerve cells to relay this information to an intergrating center
Answers: 2

Chemistry, 22.06.2019 12:20
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2
You know the right answer?
(1) A certain quantity of sodium tetraborate was weighed accurately and placed in a 250 ml volumetri...
Questions

Advanced Placement (AP), 04.11.2019 13:31

Mathematics, 04.11.2019 13:31

English, 04.11.2019 13:31

Social Studies, 04.11.2019 13:31

Mathematics, 04.11.2019 13:31


Mathematics, 04.11.2019 13:31

Mathematics, 04.11.2019 13:31



Mathematics, 04.11.2019 13:31



Biology, 04.11.2019 13:31


Advanced Placement (AP), 04.11.2019 13:31


Mathematics, 04.11.2019 13:31


English, 04.11.2019 13:31