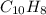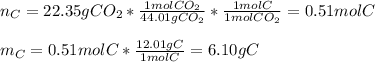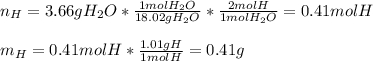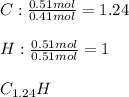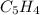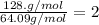Chemistry, 16.07.2021 02:30 ayoismeisjuam
6.50 g of a certain Compound x, known to be made of carbon, hydrogen and perhaps oxygen, and to have a molecular molar mass of 128. g/mol, is burned completely in excess oxygen, and the mass of the products carefully measured: product carbon dioxide water mass 22.35 g 3.66 g Use this information to find the molecular formula of x

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:10
Using complete sentences, explain how to predict the products and balance the reaction between sulfuric acid and potassium hydroxide.
Answers: 1

Chemistry, 22.06.2019 18:30
Read the claim. breakfast is an important meal. it jump starts the body’s process of using calories to break down food. appetite can decrease with age, but going too long without eating causes metabolism to slow down. current research shows that incorporating legumes such as lentils and chickpeas into meals boosts metabolism for twenty-four hours. who might benefit from this claim? people who have a fast metabolism stores that sell exercise equipment people who take vitamin supplements grocery stores that sell legumes
Answers: 1


Chemistry, 23.06.2019 04:20
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2
You know the right answer?
6.50 g of a certain Compound x, known to be made of carbon, hydrogen and perhaps oxygen, and to have...
Questions

Mathematics, 21.01.2021 23:10





Mathematics, 21.01.2021 23:10

Mathematics, 21.01.2021 23:10



History, 21.01.2021 23:10



Health, 21.01.2021 23:10


Mathematics, 21.01.2021 23:10

Mathematics, 21.01.2021 23:10

Mathematics, 21.01.2021 23:10