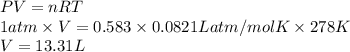Chemistry, 15.07.2021 23:20 pakabigail4796
A reaction at 5.0°C evolves 583.mmol of dinitrogen difluoride gas. Calculate the volume of dinitrogen difluoride gas that is collected. You can assume the pressure in the room is exactly 1atm . Round your answer to 3 significant digits.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Calculate the expected ph values of the buffer systems from the experiments (a,b,c,d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 01:20
Match the acid base pairs by arranging the acid name with the conjugate base formula. hydrogen carbonate hydrogen phosphate carbonic acid read water sulfuric acid phosphoric acid a. co32- b. hso4- c. hco3- d. po43- e. h2po4- f. oh-
Answers: 1


Chemistry, 22.06.2019 13:00
What happens to the average kinetic energy of a gas when the particles of the gas collide against each other at a constant temperature and volume? explain your answer.
Answers: 3
You know the right answer?
A reaction at 5.0°C evolves 583.mmol of dinitrogen difluoride gas. Calculate the volume of dinitroge...
Questions

Health, 02.09.2019 14:00





Spanish, 02.09.2019 14:00



Health, 02.09.2019 14:00

Geography, 02.09.2019 14:00


Mathematics, 02.09.2019 14:00


History, 02.09.2019 14:00



Social Studies, 02.09.2019 14:00



Chemistry, 02.09.2019 14:00

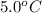 = (5 + 273) K = 278 K
= (5 + 273) K = 278 K