
Chemistry, 13.07.2021 16:30 aesthetickait
The following pairs of soluble solutions can be mixed. In some cases, this leads to the formation of an insoluble precipitate. Decide, in each case, whether or not an insoluble precipitate is formed.
a. K2S and NH4Cl
b. CaCl2 and NH4CO3
c. Li2S and MnBr2
d. Ba(NO3)2 and Ag2SO4
e. RbCO3 and NaCl

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 12:00
What is the subscript for oxygen in its molecular formula
Answers: 1

Chemistry, 22.06.2019 13:00
Asubstance is a good conductor of electricity which of the following best explains a probable position of the substance in a periodic table
Answers: 3

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 22:30
What is a number added in front of a formula in order to balance the equation
Answers: 1
You know the right answer?
The following pairs of soluble solutions can be mixed. In some cases, this leads to the formation of...
Questions

English, 22.06.2019 09:30


Chemistry, 22.06.2019 09:30

English, 22.06.2019 09:30

Business, 22.06.2019 09:30


Business, 22.06.2019 09:30







Mathematics, 22.06.2019 09:30


Social Studies, 22.06.2019 09:30


History, 22.06.2019 09:30


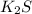 and
and 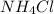 :
: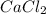 and
and 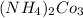 :
: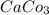 forms.
forms.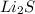 and
and 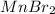 :
: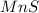 forms.
forms.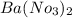 and
and 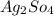 :
: 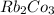 and
and 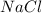 :
: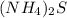 ⇒ soluble.
⇒ soluble. ⇒ soluble,
⇒ soluble, 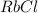 ⇒ soluble,
⇒ soluble, 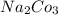 ⇒ soluble.
⇒ soluble.

