Octane (C8H18) is a component of gasoline that burns according to the following equation: C8H18(l) + 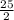 O2(g) → 8CO2(g) + 9H2O(g) ΔH∘rxn = −5074.1 kJ
What mass of octane (in g) is required to produce 8210 kJ of heat?
O2(g) → 8CO2(g) + 9H2O(g) ΔH∘rxn = −5074.1 kJ
What mass of octane (in g) is required to produce 8210 kJ of heat?
Express your answer to three significant figures.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Modern weaponry has increased the number of deaths in wars and violent conflicts.
Answers: 3

Chemistry, 22.06.2019 10:30
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1

Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

Chemistry, 22.06.2019 13:30
Which of the following has wavelength longer than the wavelength of viable light? a) x rays b) gamma rays c) radios waves d) ultraviolet waves
Answers: 1
You know the right answer?
Octane (C8H18) is a component of gasoline that burns according to the following equation: C8H18(l) +...
Questions



Social Studies, 12.11.2020 22:50



Mathematics, 12.11.2020 22:50



Computers and Technology, 12.11.2020 22:50

Health, 12.11.2020 22:50

History, 12.11.2020 22:50



History, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50

Arts, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50

Mathematics, 12.11.2020 22:50


English, 12.11.2020 22:50