
Chemistry, 13.07.2021 04:00 erikloza12pdidtx
Solutions of Cu2+ turn blue litmus red because of the equilibrium: Cu(H2O)62+(aq) + H2O(l) ↔ Cu(H2O)5(OH)+(aq) + H3O+(aq) for which Ka = 1.0 x 10-8. Calculate the pH of 0.10 M Cu(NO3)2(aq).

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:10
Stage in which a typical star has completely stopped fusion
Answers: 1

Chemistry, 22.06.2019 15:30
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins.co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2

Chemistry, 23.06.2019 05:00
How is electrolysis most commonly used to produce an energy source? a - splitting water molecules produces oxygen, which organisms breathe to fuel their bodies. b - splitting water molecules produces hydrogen gas, which is used to power machines through hydrogen fuel cells. c - splitting carbon dioxide molecules produces coal, a form of carbon that can be burned to produce heat. d - splitting carbon dioxide molecules produces natural gas, which can be burned to generate electricity in power plants.
Answers: 1
You know the right answer?
Solutions of Cu2+ turn blue litmus red because of the equilibrium: Cu(H2O)62+(aq) + H2O(l) ↔ Cu(H2O)...
Questions


Biology, 06.07.2019 03:00

Arts, 06.07.2019 03:00


Mathematics, 06.07.2019 03:00




Mathematics, 06.07.2019 03:00




Mathematics, 06.07.2019 03:00


Business, 06.07.2019 03:00


Computers and Technology, 06.07.2019 03:00

Health, 06.07.2019 03:00

History, 06.07.2019 03:00

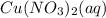 is 4.49.
is 4.49.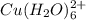 = 0.10 M
= 0.10 M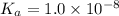
![Cu(H_{2}O)^{2+}_{6} \rightleftharpoons [Cu(H_{2}O)_{5}(OH)]^{+} + H_{3}O^{+}](/tpl/images/1393/0831/fa913.png)
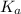 is very small. So, it is assumed that the compound will dissociate very less. Hence, x << 0.10 M.
is very small. So, it is assumed that the compound will dissociate very less. Hence, x << 0.10 M.![K_{a} = \frac{[Cu(H_{2}O)^{2+}_{6}][H_{3}O^{+}]}{[Cu(H_{2}O)^{2+}_{6}]}\\1.0 \times 10^{-8} = \frac{x \times x}{0.10}\\x = 3.2 \times 10^{-5}](/tpl/images/1393/0831/ba8e8.png)
![[H_{3}O^{+}] = 3.2 \times 10^{-5}](/tpl/images/1393/0831/f5dd0.png)
![pH = -log [H^{+}]](/tpl/images/1393/0831/8d00e.png)
![pH = -log [H^{+}]\\= - log (3.2 \times 10^{-5})\\= 4.49](/tpl/images/1393/0831/392b2.png)


