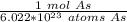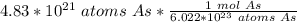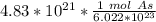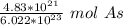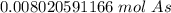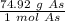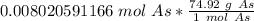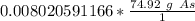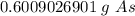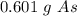Chemistry, 09.07.2021 05:20 adreyan3479
40 POINTS! WILL MARK BRAINLIEST!! Determine the mass in grams of 4.83 × 10²¹ atoms of arsenic. (The mass of one mole of arsenic is 74.92 g.)

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:10
If a molecule with a molecular formula of c13h18 is treated with an excess of h2 in the presence of finally divided pt metal under conditions required for maximum hydrogenation of the molecule to give a molecule with a formula c13h24, how many rings are in the molecule?
Answers: 3

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1

Chemistry, 23.06.2019 00:00
How do you determine the percent yield of a chemical reaction
Answers: 1

Chemistry, 23.06.2019 04:31
How does a sample of helium at 15 degree celsius compare to a sample of helium at 215 k? a) the helium at 15 degrees celsius has a higher average kinetic energy that the sample at 215 k. b) the helium at 15 degrees celsius has lower nuclear energy that the sample at 215 k. c) the helium at 15 degrees celsius has slower- moving atoms that the sample at 215 k. d) the helium at 15 degrees celsius has smaller atoms than the sample at 215 k.
Answers: 1
You know the right answer?
40 POINTS! WILL MARK BRAINLIEST!! Determine the mass in grams of 4.83 × 10²¹ atoms of arsenic. (The...
Questions

Social Studies, 17.06.2021 21:20

Social Studies, 17.06.2021 21:20




Physics, 17.06.2021 21:20


Mathematics, 17.06.2021 21:20



English, 17.06.2021 21:20

Social Studies, 17.06.2021 21:20

Mathematics, 17.06.2021 21:20

History, 17.06.2021 21:30


Mathematics, 17.06.2021 21:30


Mathematics, 17.06.2021 21:30