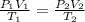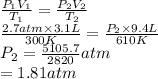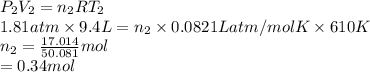A cylinder contains 3.1 L of oxygen at 300 K and 2.7 atm. The gas is heated, causing a piston in the cylinder to move outward. The heating causes the temperature to rise to 610 K and the volume of the cylinder to increase to 9.4 L.
How many moles of gas are in the cylinder?
Express your answer using two significant figures.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 12:30
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1


Chemistry, 23.06.2019 03:50
How many liters of oxygen gas, at standardtemperature and pressure, will react with 35.8 grams ofiron metal? 4 fe (s) + 3 o2 (g) → 2 fe2o3 (s)
Answers: 3
You know the right answer?
A cylinder contains 3.1 L of oxygen at 300 K and 2.7 atm. The gas is heated, causing a piston in the...
Questions








English, 24.06.2019 14:30



Mathematics, 24.06.2019 14:30

English, 24.06.2019 14:30

History, 24.06.2019 14:30



Chemistry, 24.06.2019 14:30

Chemistry, 24.06.2019 14:30



Biology, 24.06.2019 14:30

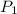 = 2.7 atm,
= 2.7 atm, 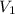 = 3.1 L,
= 3.1 L, 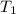 = 300 K
= 300 K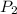 = ?,
= ?, 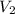 = 9.4 L,
= 9.4 L, 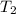 = 610 K
= 610 K