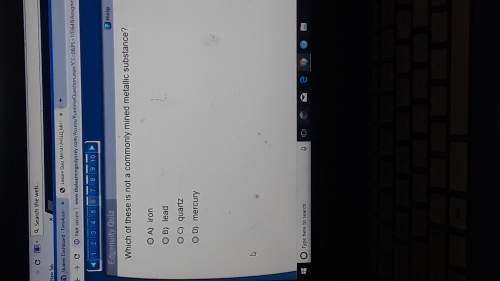
Chemistry, 07.07.2021 21:50 sawyerharper
Nitrogen gas and hydrogen gas are combined in a reaction to produce ammonia. If 6.0 moles of N2 and 6.0 moles of H2 are present in the mixture, which is the limiting reactant? Show work

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Look at the bean data from days 4–6. use these data to explain how natural selection changed the number of dark red walking beans over time. writing part
Answers: 3

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 13:30
If the concentration of phosphate in the cytosol is 2.0 mm and the concentration of phosphate in the surrounding fluid is 0.1 mm, how could the cell increase the concentration of phosphate in the cytosol? a) passive transportb) diffusionc) active transportd) osmosise) facilitated diffusion
Answers: 3

Chemistry, 22.06.2019 14:00
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3
You know the right answer?
Nitrogen gas and hydrogen gas are combined in a reaction to produce ammonia. If 6.0 moles of N2 and...
Questions


English, 04.08.2019 19:30




English, 04.08.2019 19:30

Business, 04.08.2019 19:30

History, 04.08.2019 19:30

Biology, 04.08.2019 19:30

Social Studies, 04.08.2019 19:30

Mathematics, 04.08.2019 19:30

Mathematics, 04.08.2019 19:30

Computers and Technology, 04.08.2019 19:30





History, 04.08.2019 19:30