
Chlorine can be prepared in the laboratory by the reaction of manganese dioxide with hydrochloric acid, HCl(aq) , as described by the chemical equation
MnO2(s)+4HCl(aq)⟶MnCl2(aq)+2H2O(l)+ Cl2(g)
How much MnO2(s) should be added to excess HCl(aq) to obtain 185 mL Cl2(g) at 25 °C and 745 Torr ?
mass of MnO2:

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:00
What is the molarity of 60.0 grams of naoh dissolved in 750 milliliters of water? a) 1.1 m b) 2.0 m c) 12 m d) 75 m
Answers: 1

Chemistry, 22.06.2019 10:20
Gwhich r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? which r group would most likely be found in a hydrophobic area of the tertiary structure of a globular protein? −ch2−oh −ch2−o||c−nh2 −ch2−coo− −ch2−ch2−ch2−ch2−n+h3
Answers: 3


Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2
You know the right answer?
Chlorine can be prepared in the laboratory by the reaction of manganese dioxide with hydrochloric ac...
Questions

English, 10.05.2021 20:50

Physics, 10.05.2021 20:50

Mathematics, 10.05.2021 20:50





Social Studies, 10.05.2021 20:50






Chemistry, 10.05.2021 20:50



Geography, 10.05.2021 20:50



Arts, 10.05.2021 21:00

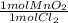 = 0.00696 mol MnO₂
= 0.00696 mol MnO₂

