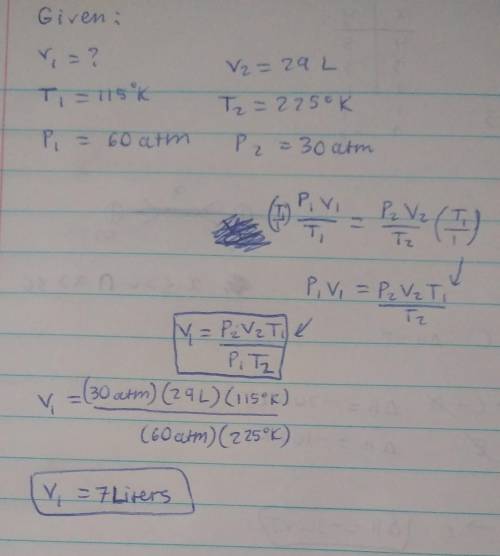Chemistry, 05.07.2021 20:10 alyssalefeber
I have an unknown volume of gas held at a temperature of 115 K in a container with a pressure of 60atm. If by increasing the temperature to 225 K and decreasing the pressure to 30. atm causes the volume of the gas to be 29 liters, how many liters of gas did I start with? SHOW YOUR WORK

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:10
What can be added to the examples section of each circle? endothermic: ice melting into water, and a heat pack becoming warm exothermic: a glow stick glowing, and fireworks exploding endothermic: ice melting into water, and an instant ice pack turning cold exothermic: fireworks exploding, and gasoline burning endothermic: a glow stick glowing, and a heat pack becoming warm exothermic: an instant ice pack turning cold, and ice melting into water endothermic: gasoline burning, and an instant ice pack turning cold exothermic: ice melting into water, and an instant ice pack turning cold
Answers: 1


Chemistry, 22.06.2019 11:40
Consider this equilibrium: n29) + o2(g) + 2no(c).nitrogen gas and oxygen gas react when placed in a closed container. as the reaction proceeds towards equilibrium, what happens to the rate of thereverse reaction?
Answers: 1

You know the right answer?
I have an unknown volume of gas held at a temperature of 115 K in a container with a pressure of 60a...
Questions



Chemistry, 27.01.2020 10:31

Social Studies, 27.01.2020 10:31





Chemistry, 27.01.2020 10:31


History, 27.01.2020 10:31


Biology, 27.01.2020 10:31

Computers and Technology, 27.01.2020 10:31

Mathematics, 27.01.2020 10:31




Mathematics, 27.01.2020 10:31

Mathematics, 27.01.2020 10:31