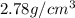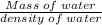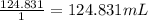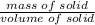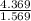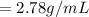A 4.369 g sample of metal is placed in a flask. Water is added to the flask and the total volume in the flask is read to be 126.4 ml. The mass of the water, flask, and metal is 268.5 g. If the mass of the flask is 139.3 g and the density of water is 1.000 g/mL, the density of the solid is g/cm3.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1

Chemistry, 22.06.2019 09:20
What will most likely happen when two bromine atoms bond together?
Answers: 3

Chemistry, 22.06.2019 11:40
Which of these expressions are correct variations of the combined gas law? p1v1t2 = p2v2t1 both
Answers: 2

Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
You know the right answer?
A 4.369 g sample of metal is placed in a flask. Water is added to the flask and the total volume in...
Questions


English, 20.10.2019 16:10

Mathematics, 20.10.2019 16:10

Biology, 20.10.2019 16:10


Chemistry, 20.10.2019 16:10

English, 20.10.2019 16:10

English, 20.10.2019 16:10

Mathematics, 20.10.2019 16:10


English, 20.10.2019 16:10





Mathematics, 20.10.2019 16:10

Mathematics, 20.10.2019 16:10