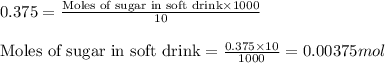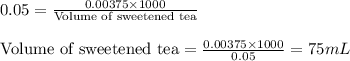Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:50
If the mass of the products measured 120g what would the mass of the reactants a. 30g b. 60g c. 120g d. 240g
Answers: 1

Chemistry, 21.06.2019 22:30
Determine the wavelength of the light absorbed when an electron in a hydrogen atom makes a transition from an orbital in the n=3 level to an orbital in the n=7 level.
Answers: 2

Chemistry, 23.06.2019 06:00
Robert leaves a chocolate bar in his car while attending school all day. when he goes to his car in the afternoon, the bat has changed into gooey liquid. what happened to the chocolate bar
Answers: 1

Chemistry, 23.06.2019 09:20
Asolution of naoh has a concentration of 25.00% by mass. what mass of naoh is present in 0.250 g of this solution? use the periodic table in the toolbar if needed. 0.0625 g what mass of naoh must be added to the solution to increase the concentration to 30.00% by mass? g
Answers: 2
You know the right answer?
What volume (mL) of the sweetened tea described in Example 1 contains the same amount of sugar (mol)...
Questions

Mathematics, 10.07.2019 10:00


Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00


History, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00



Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00


Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00

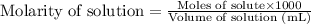 .....(1)
.....(1)