Chemistry, 28.06.2021 15:40 negativechill
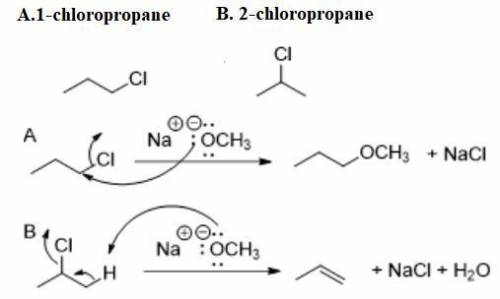
Compound A and compound B are constitutional isomers with molecular formula C3H7Cl. When compound A is treated with sodium methoxide, a substitution reaction predominates. When compound B is treated with sodium methoxide, an elimination reaction predominates.
Required:
Propose structures A and B.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:00
Suppose the universe were completely empty except for one object-a solid sphere moving through space of 100 km/s. what sort of path would the object be moving in? explain your answer
Answers: 1

Chemistry, 22.06.2019 10:00
Which sentence about particles in matter is true? a. atoms are present in solids and liquids but not in gases. b. the particles of matter are in constant motion. c. the same kinds of atoms are found in different elements. d. when a solid changes to a liquid, the sizes of the particles change.
Answers: 1

Chemistry, 22.06.2019 16:00
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1
You know the right answer?
Compound A and compound B are constitutional isomers with molecular formula C3H7Cl. When compound A...
Questions

Mathematics, 27.06.2019 15:50

English, 27.06.2019 15:50

English, 27.06.2019 15:50


Computers and Technology, 27.06.2019 15:50


Mathematics, 27.06.2019 15:50

Mathematics, 27.06.2019 15:50




Biology, 27.06.2019 15:50

Social Studies, 27.06.2019 15:50

History, 27.06.2019 15:50

English, 27.06.2019 15:50

Mathematics, 27.06.2019 15:50

English, 27.06.2019 15:50

English, 27.06.2019 15:50


English, 27.06.2019 15:50