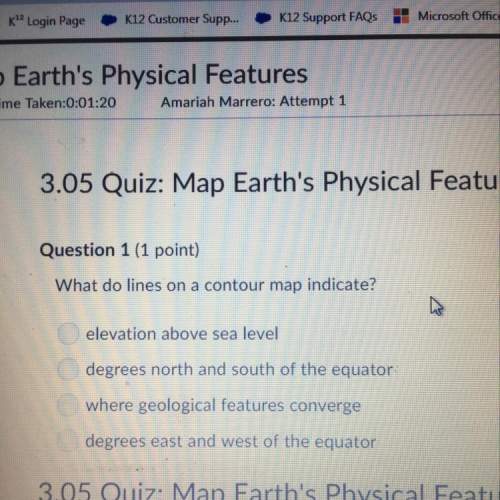
Chemistry, 26.06.2021 09:40 ashl3yisbored
Use the periodic table to determine the electron configuration for aluminum (AI) and arsenic (As) in
noble-gas notation
AL
o [Ar]3s23p!
lo (He]2s22p63s23p!
0 [Ne]3s23p!
0 [Ne]3s23p2
DONE

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:00
In a sample of oxygen gas at room temperature, the average kinetic energy of all the balls stays constant. which postulate of kinetic molecular theory best explains how this is possible? a. attractive forces between gas particles are negligible because the particles of an ideal gas are moving so quickly. b. collisions between gas particles are elastic; there is no net gain or loss of kinetic energy. c. gases consist of a large number of small particles, with a lot of space between the particles. d. gas particles are in constant, random motion, and higher kinetic energy means faster movement.
Answers: 1

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 18:00
Many pharmaceutical drugs are organic compounds that were originally synthesized in the laboratory. which two scientific disciplines are bridged by pharmaceutical drugs? chemistry and forensics chemistry and medicine biology and forensics biology and criminology
Answers: 2

You know the right answer?
Use the periodic table to determine the electron configuration for aluminum (AI) and arsenic (As) in...
Questions


English, 07.01.2020 17:31

Social Studies, 07.01.2020 17:31


History, 07.01.2020 17:31



English, 07.01.2020 17:31

History, 07.01.2020 17:31


Mathematics, 07.01.2020 17:31



Mathematics, 07.01.2020 17:31


History, 07.01.2020 17:31

English, 07.01.2020 17:31

Social Studies, 07.01.2020 17:31


World Languages, 07.01.2020 17:31