
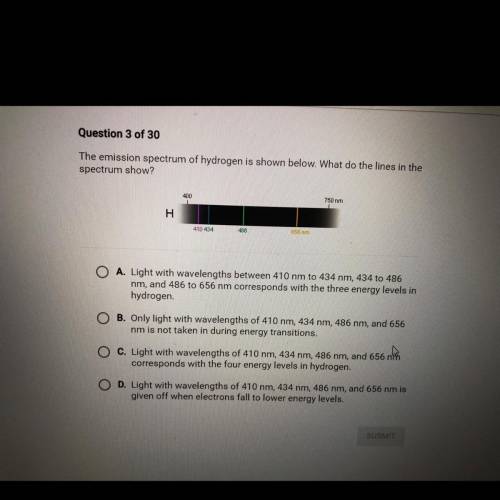
The emission spectrum of hydrogen is shown below. What do the lines in the
spectrum show?
A. Light with wavelengths between 410 nm to 434 nm, 434 to 486 nm, and 486 to 656 nm corresponds with the three energy levels in
hydrogen
B. Only light with wavelengths of 410 nm, 434 nm, 486 nm, and 656 nm is not taken in during energy transitions.
C. Light with wavelengths of 410 nm, 434 nm, 486 nm, and 656 no corresponds with the four energy levels in hydrogen.
D. Light with wavelengths of 410 nm, 434 nm, 486 nm, and 656 nm is given off when electrons fall to lower energy levels.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 07:30
According to the vsepr theory what is the shape of a molecule that has a central atom valence three other items with no lone pairs of electrons
Answers: 1

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

Chemistry, 22.06.2019 11:50
Calculate the molarity of each of the following solutions. part a) 0.12 mol of lino3 in 5.5 l of solution part b) 60.7 g c2h6o in 2.48 l of solution part c) 14.2 mg ki in 100 ml of solution
Answers: 2

Chemistry, 23.06.2019 02:00
Anitrogen atom and an oxygen atom combine chemically to form nitric oxide. what is nitric oxide?
Answers: 1
You know the right answer?
The emission spectrum of hydrogen is shown below. What do the lines in the
spectrum show?
Questions

Mathematics, 21.01.2020 10:31


History, 21.01.2020 10:31

History, 21.01.2020 10:31

World Languages, 21.01.2020 10:31

Arts, 21.01.2020 10:31



Mathematics, 21.01.2020 10:31


Mathematics, 21.01.2020 10:31

Mathematics, 21.01.2020 10:31

Advanced Placement (AP), 21.01.2020 10:31

Mathematics, 21.01.2020 10:31

Mathematics, 21.01.2020 10:31

Mathematics, 21.01.2020 10:31

Health, 21.01.2020 10:31



History, 21.01.2020 10:31



