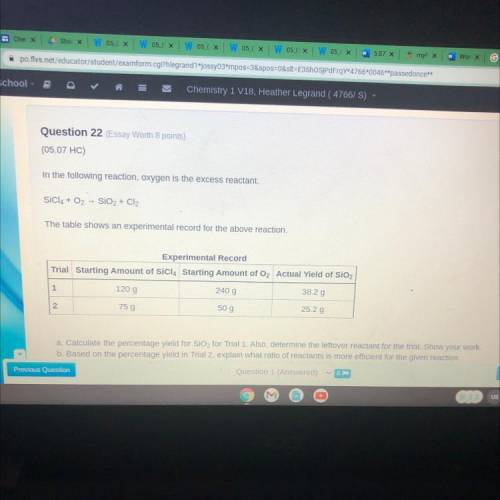
Question 22 (Essay Worth 8 points)
(05.07 HC)
In the following reaction, oxygen is the excess...

Question 22 (Essay Worth 8 points)
(05.07 HC)
In the following reaction, oxygen is the excess reactant.
SÍCIA + O2 - SiO2 + Cl2
The table shows an experimental record for the above reaction.
Experimental Record
Trial Starting Amount of SiCl4 Starting Amount of O2 Actual Yield of SiO2
1
120 g
240 g
38.2 g
N
75 g
50 g
25.2 g
a. Calculate the percentage yield for SiO2 for Trial 1. Also, determine the leftover reactant for the trial. Show your work
b. Based on the percentage yield in Trial 2, explain what ratio of reactants is more efficient for the given reaction.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 08:30
In the reaction between a crushed antacid tablet and vinegar what gas is emitted
Answers: 2

Chemistry, 22.06.2019 23:30
If it is an isoelectronic series select true, if not select false. o2-, s2-, se2-, te2- na+, k+, rb+, cs+ n3-, p3-, as3-, sb3- ag, cd+, sn3+, sb4+ f-, cl-, br-, i- f-, ne, na+, mg2+ s2-, s, s6+
Answers: 1

Chemistry, 23.06.2019 00:00
How many peaks will be present in a mass spectrum for brcl?
Answers: 1
You know the right answer?
Questions

History, 13.09.2020 21:01

Spanish, 13.09.2020 21:01

Mathematics, 13.09.2020 21:01

History, 13.09.2020 21:01

Mathematics, 13.09.2020 21:01

Mathematics, 13.09.2020 22:01

Biology, 13.09.2020 22:01

English, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

History, 13.09.2020 22:01

Geography, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Spanish, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Health, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01

Mathematics, 13.09.2020 22:01



