
Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1


Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1

Chemistry, 22.06.2019 22:30
Molecular iodine, i2(g), dissociates into iodine atoms at 625 k with a first-order rate constant of 0.271 s−1. part a part complete what is the half-life for this reaction?
Answers: 3
You know the right answer?
Geef de systematische namen van SiF2...
Questions


Mathematics, 05.11.2020 23:40


Social Studies, 05.11.2020 23:40



German, 05.11.2020 23:40

Mathematics, 05.11.2020 23:40

Mathematics, 05.11.2020 23:40



Mathematics, 05.11.2020 23:40

Mathematics, 05.11.2020 23:40

Mathematics, 05.11.2020 23:40




Health, 05.11.2020 23:40

Biology, 05.11.2020 23:40

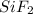 .
. 

