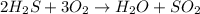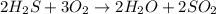A student wrote a chemical equation as shown.
2H₂S+ 302 H₂O + SO₂
What should the stude...

Chemistry, 20.06.2021 07:20 EatsChiken
A student wrote a chemical equation as shown.
2H₂S+ 302 H₂O + SO₂
What should the student do to balance the equation?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 14:30
Which is true of the reactants in this displacement reaction? fe + 2hcl fecl2 + h2 a. the reactants are located to the left of the arrow in the chemical equation. b. the reactants contain 1 iron atom, 2 hydrogen atoms, and 1 chlorine atom. c. the reactants are the atoms, molecules, or compounds formed in the reaction. d. the reactants have the same physical and chemical properties as the products.
Answers: 1

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3

Chemistry, 22.06.2019 13:50
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1
You know the right answer?
Questions



Mathematics, 02.09.2019 11:10

History, 02.09.2019 11:10

Mathematics, 02.09.2019 11:10


Biology, 02.09.2019 11:10



History, 02.09.2019 11:10


History, 02.09.2019 11:10


Biology, 02.09.2019 11:10

Mathematics, 02.09.2019 11:10

Social Studies, 02.09.2019 11:10




Mathematics, 02.09.2019 11:10

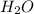 and
and 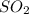 to make the equation balanced.
to make the equation balanced.