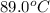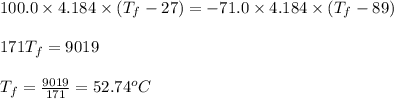Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Which organic molecule is found in the chromatin of cells?
Answers: 1


Chemistry, 21.06.2019 23:00
What is the molecular formula for a compound that is 46.16% carbon, 5.16% hydrogen, and 48.68% fluorine? the molar mass of the compound is 156.12 g/mol
Answers: 2

Chemistry, 22.06.2019 04:30
Acamcorder has a power rating of 17 watts. if the output voltage from its battery is 7 volts, what current does it use?units:
Answers: 1
You know the right answer?
A 100.0-g sample of water at 27.0oC is poured into a 71.0-g sample of water at 89.0oC. What will be...
Questions


Business, 07.03.2021 21:20






History, 07.03.2021 21:20

Social Studies, 07.03.2021 21:20

Biology, 07.03.2021 21:20








English, 07.03.2021 21:20

Mathematics, 07.03.2021 21:20

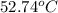
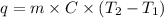
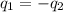
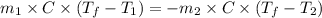 ......(1)
......(1)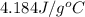
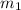 = mass of water of sample 1 = 100.0 g
= mass of water of sample 1 = 100.0 g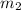 = mass of water of sample 2 = 71.0 g
= mass of water of sample 2 = 71.0 g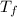 = final temperature of the system = ?
= final temperature of the system = ?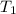 = initial temperature of water of sample 1 =
= initial temperature of water of sample 1 = 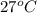
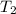 = initial temperature of the water of sample 2 =
= initial temperature of the water of sample 2 =