
Chemistry, 19.06.2021 04:20 zanaplen27
PLEASE HELP QUICK
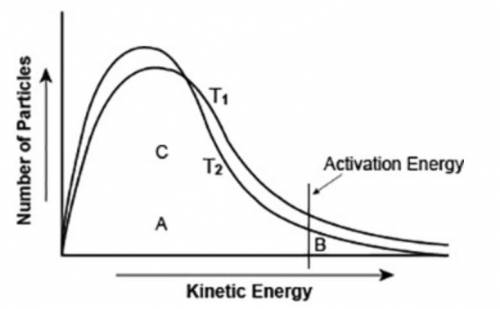
The graph shows the distribution of energy in the particles of two gas samples at different temperatures, T1 and T2. A, B, and C represent individual particles.
Based on the graph, which of the following statements is likely to be true? (3 points)
Particle A is more likely to participate in the reaction than particle B.
Particle C is more likely to participate in the reaction than particle B.
The number of particles able to undergo a chemical reaction is less than the number that is not able to.
More gas particles participate in the reaction at T2 than at T1.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 21:10
Harvey mixes two liquids. which observation of the new mixture most likely indicates a precipitate is forming?
Answers: 2


Chemistry, 22.06.2019 10:30
Use this information to determine the number of calends electrons in the atoms. which of the following correctly compares the stability of the two atoms? a) both are unreactive b) both are highly reactive c) a is unreactive and d is reactive d) a is reactive and d is unreactive
Answers: 2

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3
You know the right answer?
PLEASE HELP QUICK
The graph shows the distribution of energy in the particles of two gas samples at...
Questions

Mathematics, 17.12.2021 03:20

Mathematics, 17.12.2021 03:20

Mathematics, 17.12.2021 03:20







Social Studies, 17.12.2021 03:20



Mathematics, 17.12.2021 03:30


Mathematics, 17.12.2021 03:30







