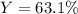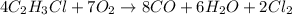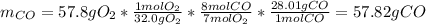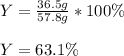Chemistry, 18.06.2021 02:50 sadsociety41
For the following reaction: 4C2H3Cl + 702 → 8CO + 6H20 + 2Cl2 If the reaction of 57.8 grams of O2 produces 36.5 grams of CO, what is the percent yield?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:00
The compound methyl butanoate smells like apples. its percent composition is 58.8% c, 9.9% h, and 31.4% o. what’s the empirical formula ?
Answers: 1

Chemistry, 22.06.2019 00:30
You have 125g of a certain seasoning and are told that it contains 76.0 g of salt what is the percentage of salt by mass in this seasoning
Answers: 1

Chemistry, 22.06.2019 05:40
Why did southern business leaders want to increase the number of slaves
Answers: 1

Chemistry, 22.06.2019 09:40
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3
You know the right answer?
For the following reaction: 4C2H3Cl + 702 → 8CO + 6H20 + 2Cl2 If the reaction of 57.8 grams of O2 pr...
Questions

History, 23.09.2019 03:30


Chemistry, 23.09.2019 03:30

Physics, 23.09.2019 03:40





World Languages, 23.09.2019 03:50



Mathematics, 23.09.2019 03:50

History, 23.09.2019 03:50

Mathematics, 23.09.2019 03:50

Mathematics, 23.09.2019 03:50


Business, 23.09.2019 03:50

English, 23.09.2019 03:50