
Chemistry, 16.06.2021 20:20 PONBallfordM89
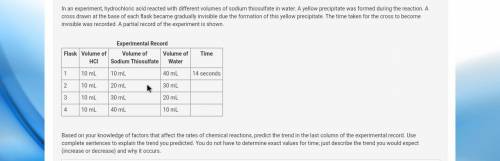
Based on your knowledge of factors that affect the rates of chemical reactions, predict the trend in the last column of the experimental record. Use complete sentences to explain the trend you predicted. You do not have to determine exact values for time; just describe the trend you would expect (increase or decrease) and why it occurs.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 22:00
The graph above shows how the price of cell phones varies with the demand quantity. the equilibrium price for cell phones is where both supply and demand quantities equal $100, 5,000 5,000, $100
Answers: 2

Chemistry, 22.06.2019 15:00
‘which reaction would most likely require the use of an inert electrode?
Answers: 1


Chemistry, 23.06.2019 03:00
The size (radius) of an oxygen molecule is about 2.0 ×10−10m. make a rough estimate of the pressure at which the finite volume of the molecules should cause noticeable deviations from ideal-gas behavior at ordinary temperatures (t= 300k ). assume that deviatons would be noticeable when volume of the gas per molecule equals the volume of the molecule itself.
Answers: 3
You know the right answer?
Based on your knowledge of factors that affect the rates of chemical reactions, predict the trend in...
Questions

History, 26.08.2020 22:01

Mathematics, 26.08.2020 22:01






English, 26.08.2020 22:01

History, 26.08.2020 22:01

English, 26.08.2020 22:01


English, 26.08.2020 22:01

English, 26.08.2020 22:01

Mathematics, 26.08.2020 22:01




English, 26.08.2020 22:01

History, 26.08.2020 22:01



