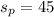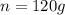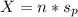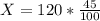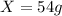Chemistry, 14.06.2021 16:40 LoserMcBadface
A mixture of coarse sand and sugar is 45.0 percent sand by mass. 120.0 grams (g) of the mixture is placed in a fine-mesh cloth bag and dunked repeatedly in Lake Michigan. After drying, the mass of the contents of the bag equals: .
A. 66.0 g
B. 120.0 g
C. 65.0 g
D. 72.00 g
E. 54.0 g

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 22:40
Covalent bonds generally form when the bonded elements have a difference in electronegativity less than 1.5. subtract the electronegativities for the following pairs of elements and predict whether they form a covalent bond. electronegativity difference of c and c: ionic covalent electronegativity difference of mg and cl: ionic covalent
Answers: 1

Chemistry, 23.06.2019 02:00
What can be done to make a solid solute dissolve faster in a liquid solvent?
Answers: 1

Chemistry, 23.06.2019 03:50
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer
Answers: 1

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
A mixture of coarse sand and sugar is 45.0 percent sand by mass. 120.0 grams (g) of the mixture is p...
Questions




Arts, 08.11.2019 00:31

Mathematics, 08.11.2019 00:31

History, 08.11.2019 00:31

Mathematics, 08.11.2019 00:31


Physics, 08.11.2019 00:31

Mathematics, 08.11.2019 00:31



Health, 08.11.2019 00:31

Mathematics, 08.11.2019 00:31



Mathematics, 08.11.2019 00:31


History, 08.11.2019 00:31