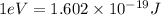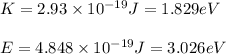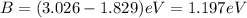(A) Calculate the wavelength (in nm) of light with energy 1.89 × 10–20 J per photon, (b) For light of wavelength 410 nm, calculate the number of photons per joule, (c) Determine the binding energy (in eV) of a metal if the kinetic energy possessed by an ejected electron [using one of the photons in part (b)] is 2.93 × 10–19 J.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:00
Which of the following statements is true? question 4 options: nuclear decay rates vary with the conditions of the reaction, but chemical reaction rates do not. chemical reaction rates vary with the conditions of the reaction, but nuclear decay rates do not. neither chemical reaction rates nor nuclear decay rates vary with the conditions of the reaction. both chemical reaction rates and nuclear decay rates vary with the conditions of the reaction.
Answers: 1

Chemistry, 22.06.2019 00:00
Several kinds of bears are found on earth. most bears are brown or black, but one type of bear, the polar bear, is white. what process led to this difference in fur color? explain your answer.
Answers: 1

Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1

You know the right answer?
(A) Calculate the wavelength (in nm) of light with energy 1.89 × 10–20 J per photon, (b) For light o...
Questions

Health, 06.01.2020 18:31


Mathematics, 06.01.2020 18:31


Business, 06.01.2020 18:31



Computers and Technology, 06.01.2020 18:31

Social Studies, 06.01.2020 18:31



Computers and Technology, 06.01.2020 18:31

Business, 06.01.2020 18:31



Social Studies, 06.01.2020 18:31


Social Studies, 06.01.2020 18:31


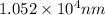
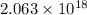
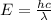 ......(1)
......(1)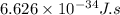
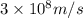
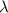 = wavelength
= wavelength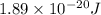
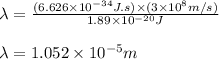
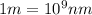
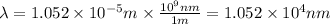
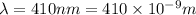
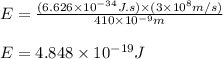
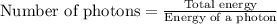
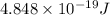
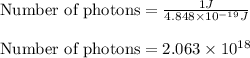
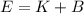 .....(2)
.....(2)