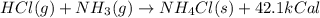Chemistry, 12.06.2021 23:40 kenisonpaigebosma
When HCl(g) reacts with NH3(g) to form NH4Cl(s), 42.1 kcal of energy are evolved for each mole of HCl(g) that reacts. Write a balanced equation for the reaction with an energy term in kcal as part of the equation.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Which of the following best explains why the end of a spoon sticking out of a cup of hot water also gets hot? question 7 options: the heat from the hot water is conducted through the spoon handle the hot water heats the air surrounding the upper part of the spoon. the hot water causes a physical change in the spoon handle. the hot water causes a chemical reaction to take place in the spoon.
Answers: 2

Chemistry, 22.06.2019 23:00
What is the most common reason for matter changing its state?
Answers: 1

Chemistry, 23.06.2019 05:00
What is dhmo? hint: you find it everywhere something is wet..
Answers: 1

Chemistry, 23.06.2019 05:00
Which of the following describes qualitative data? a) recording the temperature of a solid as it is warmed. b) noting the color of a solution as it is heated. c) measuring the volume of an object by water displacement. d) taking the mass of an object using a balance.
Answers: 2
You know the right answer?
When HCl(g) reacts with NH3(g) to form NH4Cl(s), 42.1 kcal of energy are evolved for each mole of HC...
Questions

Social Studies, 11.12.2020 03:40


Mathematics, 11.12.2020 03:40







Mathematics, 11.12.2020 03:40





Chemistry, 11.12.2020 03:40



History, 11.12.2020 03:40

Computers and Technology, 11.12.2020 03:40

History, 11.12.2020 03:40